Tag
vaccine
-
Why reporters should cover this week’s CDC meeting on hepatitis B vaccine and more
A CDC committee will discuss its current policy of universal hepatitis B vaccination at birth and other recommendations from Dec.…

-
What to know about Robert F. Kennedy Jr.’s anti-vaccine advocacy
Audiences need ongoing, accurate information about Kennedy’s beliefs on vaccines and health policy — and what he could do as…

-
How to report on the COVID-19 uptick when the public says ‘meh’
COVID-19 cases have begun rising again as the new school year looms. Here are some fresh angles for covering the…

-
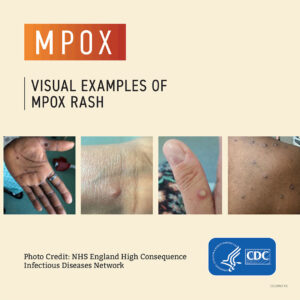
The threat of mpox returns: How reporters should cover the disease
Mpox, formerly known as monkeypox, could make a resurgence this summer in the US. What reporters need to know.

-
Biden’s bid for a nursing home vaccine mandate faces obstacles
EDITOR’S NOTE: This is the second in a series about the Biden Administration’s efforts to increase COVID-19 vaccination rates among…

-
Federal funds at stake as Biden Administration seeks to require vaccines for nursing home workers
Is a new push by the Biden Administration to increase COVID-19 vaccinations among nursing home workers the right approach? While…

-
Pfizer lays out timeline for its COVID-19 vaccine and a new flu vaccine
In a teleconference announcing the company’s first-quarter 2021 earnings earlier this month, Pfizer laid out its timeline for when different…

-
One journalist’s efforts to counter misinformation about COVID-19 vaccines
Alarm over the impact of COVID-19 misinformation has been growing, especially with increasing efforts by right-wing groups to spread misinformation…

-
Vaccine hesitancy among rural elders, caregivers nearly double of more urban counterparts
A recent survey of family caregivers revealed some troubling information about the divide between rural and urban communities regarding COVID-19…

-
If AstraZeneca’s vaccine is authorized, be prepared to write those explainers
Just a day after AstraZeneca announced long-awaited interim results from its U.S. phase 3 trial earlier this week, the company…