Tag
pharmaceutical industry
-
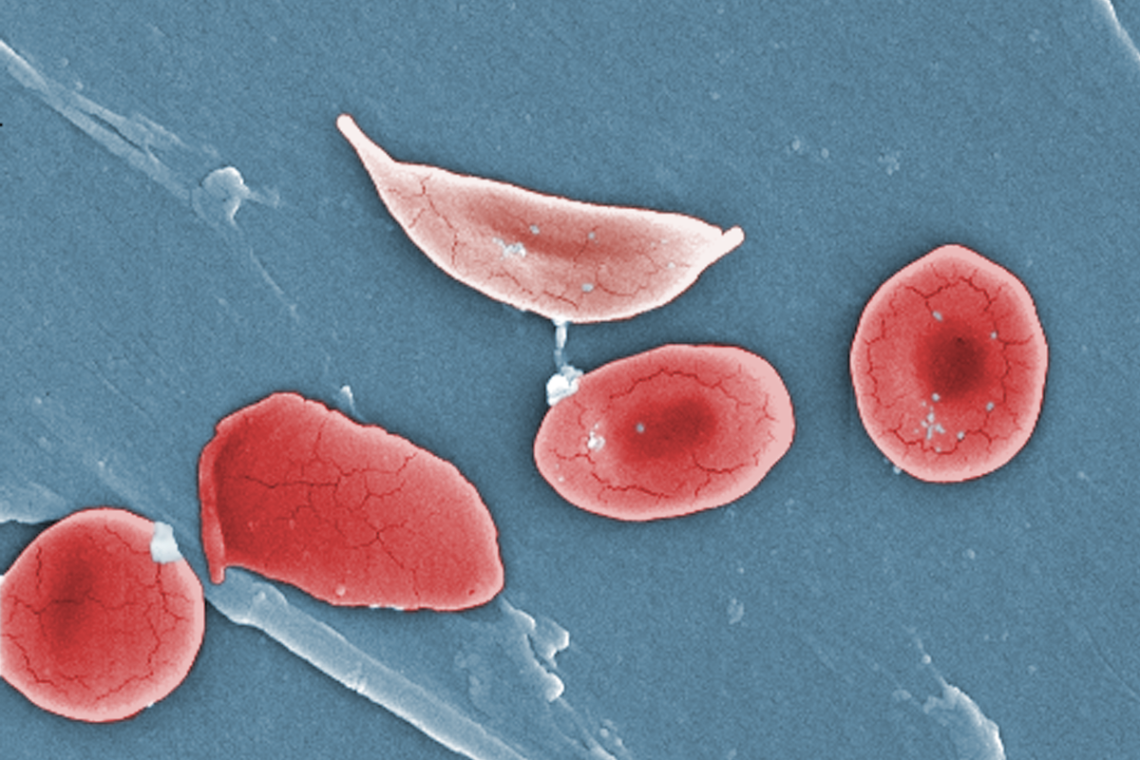
A sickle cell drug that was hyped in headlines is now suspected of harming patients
The abrupt recall of a new treatment for sickle cell disease serves as a reminder of why journalists should avoid…

-
Despite transparency, doctors accept billions from drug and device industry
This finding is a reminder for journalists to inquire whether clinician sources have financial disclosures that might bias their views.

-
When reporting on efforts to reduce drug prices, be skeptical and follow the money
The Trump administration has been talking tough on drug prices for many months and, of course, pharmaceutical companies and other…

-
Webcast to feature author of book on rampant fraud in the generic drug industry
Investigative journalist Katherine Eban will discuss her new book documenting rampant fraud in the generic drug industry during an AHCJ…

-
Do you have all the evidence on a drug that you’re reporting about?
The registration and reporting requirements of Clinicaltrials.gov are vital to informing the evidence base – but only if study sponsors…

-
•
Reporters struggle to learn the facts after only three get briefing on drug price proposals
When newly installed Health and Human Services Secretary Alex Azar held one of his first meetings with the media on…

-
•
D.C. journalists gather, meet with pharmaceutical representatives
About 25 journalists gathered on March 18 at Bistro d’Oc in Washington, D.C., for an AHCJ chapter happy hours event…

-
•
Experts share realities behind generic, specialty drug pricing
Health journalists received a few lessons in economics during a discussion last week on some alarming drug trends – largely…

-
•
Prescribing data and the side effects of assumptions #ahcj14
Reporters curious about the financial relationship between physicians and pharmaceutical companies can use publicly available data as a starting point…

-
•
Reporter focuses on chronic pain for series on opioid use
When The Cincinnati Enquirer set out to look at the societal costs of the deadly opioid crisis, reporter Lisa Bernard-Kuhn…