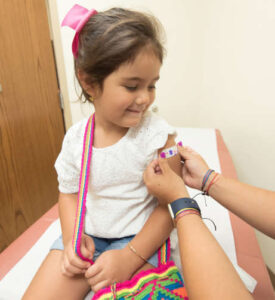
Last week Pfizer and BioNTech announced promising safety and effectiveness data for use of its COVID-19 vaccine in children ages 5 to 11, setting a potential path for ending the pandemic.
“It won’t be a silver bullet, but [vaccines for kids] will be a step in the right direction,” Katelyn Jetelina, an epidemiologist at the University of Texas Health Science Center at Houston, wrote in her newsletter called “Your Local Epidemiologist.”
On Sept. 20, the companies said a trial of 2,268 children ages 5 to 11, showed a “robust” neutralizing antibody response, using a 2-dose regimen, administered 3 weeks apart. The dose of the vaccine was lower (10 micrograms) than what is given to those 12 and older (30 micrograms) because it produced fewer side effects and still resulted in a strong immune response.
An important caveat is that there was no data on how much the vaccine reduced the chance that a child would become sick because there weren’t illnesses in kids to provide that assessment, according to STAT News. However, experts told STAT that given the antibody levels the vaccine produces, it was “reasonable to assume” that protection from illness would be comparable to the protection in children 12 and older.
“It’s not a lot to go on, but what we do have to go on looks great,” Kathleen Neuzil, director of the Center for Vaccine Development and Global Health at the University of Maryland School of Medicine told STAT. “A lower dose of this vaccine in children appears as good as the higher dose in older children and adults.”
Pfizer and BioNTech intend to apply for emergency use authorization of the vaccine with the Food and Drug Administration by the end of September, paving the way for kids to be vaccinated by Halloween. Data on a vaccine trial for kids ages 6 months to 4 years old won’t be available until the end of the year, a Pfizer official told The New York Times.
After the companies file their request for an emergency use authorization, the next step for journalists to watch for is an FDA advisory committee meeting assessing the data. The group will then make a recommendation to the agency. If approved, the FDA would amend a previous authorization for children ages 12 to 15. About a week later, the CDC would then consider the application, and if approved, set the national guidelines for the vaccine’s use. Then vaccinations could begin.
Jetelina noted it’s possible the FDA could determine there still isn’t enough data to approve the emergency use authorization because of concern about myocarditis, an inflammation of the heart muscle. Pfizer said there were no cases of myocarditis in the trial, but the study wasn’t large enough for a case of myocarditis to arise. Data from vaccinations for those 12 and older is showing that around 75 to 100 mild cases of myocarditis are occurring for every 1 million young males who are vaccinated.
The FDA will have to determine if, despite the lack of data, the benefit of reducing hospitalizations and deaths outweighs the risks of myocarditis, she said. As of Sept. 16, children represented 25.7% of new COVID-19 cases in the past week, according to the American Academy of Pediatrics.
Experts to call
Health care reporters interested in this story can contact the following experts:
- Katelyn Jetelina, Ph.D., M.P.H, assistant professor of epidemiology, human genetics and environmental sciences at the University of Texas Health Science Center at Houston. Email: katelyn.k.jetelina@uth.tmc.edu
- Yvonne Maldonado, M.D., who led the Pfizer children’s trial at Stanford University and chairs the American Academy of Pediatrics’ infectious disease committee. Email: bonniem@stanford.edu
- Kathleen Neuzil, M.D., M.P.H., F.I.D.S.A., director of the Center for Vaccine Development and Global Health at the University of Maryland School of Medicine. Email: kneuzil@som.umaryland.edu
- Flor M. Munoz, M.D., M.Sc., F.A.A.P, an associate professor of pediatrics at Baylor College of Medicine, and principal investigator in the Pfizer trial at Baylor. Email: florm@bcm.edu
- Paul Offit, M.D., director of the Vaccine Education Center and a professor of pediatric infectious disease at Children’s Hospital of Philadelphia, and member of the FDA’s Vaccines and Related Biological Products Advisory Committee. Email: Offit@email.chop.edu
- Andrew T. Pavia, M.D., the chief of the division of pediatric infectious disease at the University of Utah and former member of the U.S. Health and Human Services Vaccine Safety Working Group. Email: andy.pavia@hsc.utah.edu




