Tag
physician payments
-
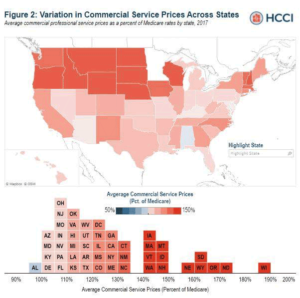
Report shows wide variation in what insurers pay providers versus what Medicare would have paid
During the coronavirus pandemic, any number of good news stories seem to get little or no coverage because most health…

-
•
CMS data should give reporters a plethora of new stories to cover
As Charles Ornstein pointed out, the Centers for Medicare and Medicaid Services announced that it will release payment information for individual physicians…

-
•
AHCJ calls for accessible reporting of physician payments
The Centers for Medicare & Medicaid Services should create an easily usable and searchable database when it publishes information from…