Tag
kaiser health news
-
How a press release sparked a storyabout critical services for sexual assault survivors
Soon after Arielle Zionts, a rural health reporter for Kaiser Health News, read a press release about telehealth services for…

-
Journalist offers tips for investigating private equity firms
Editor’s note: This is the second of two posts on covering private equity.Lack of transparency is a giant hurdle in…

-
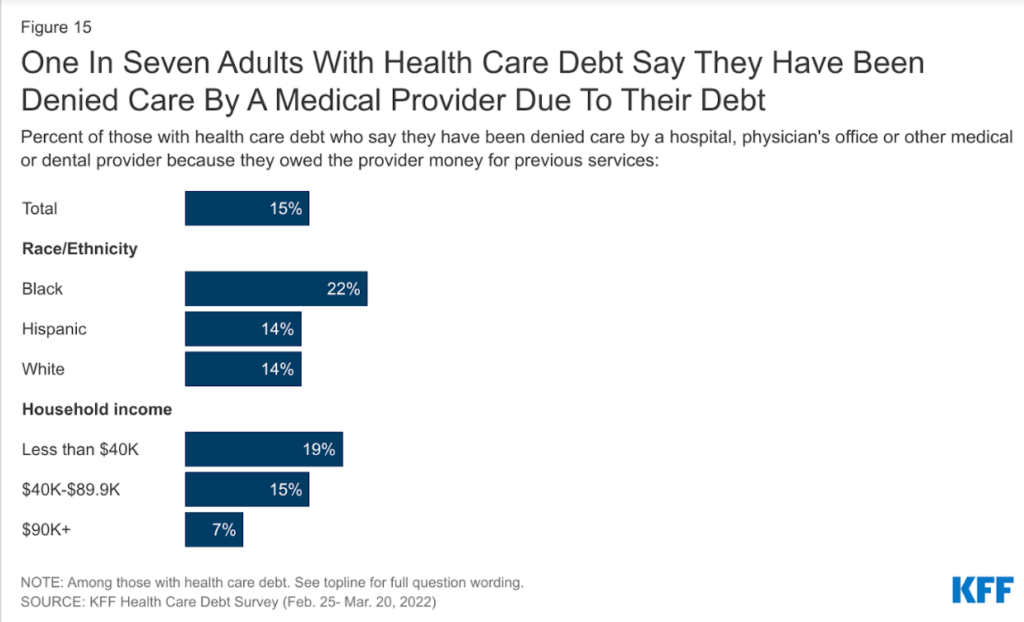
Why it’s important to cover hospitals suing patients over medical debt
Recent news shows the need for health care journalists to investigate the aggressive measures hospitals use when patients can’t pay…

-
4 ways to ramp up your reporting on rural hospitals
When reporting on the state of rural medical facilities, there’s more to the story than the demand for health care…

-
•
FDA approval causes drug price to skyrocket
In a collaboration between The Philadelphia Inquirer and Kaiser Health News, Harris Meyer looks at the case of colchicine, a…

-
Post-reform plans may still be too spendy for some
Kaiser Health News’ Jordan Rau reports that, even if current health care proposals (Rau focuses on the House bill) were…

-
•
Report looks at nonprofits’ health reporting
Maralee Schwartz, a Shorenstein Center Fellow at Harvard University, has written a report titled “Getting It for Free: When Foundations…

-
•
Galewitz takes job with Kaiser Health News
Phil Galewitz, a health writer at The Palm Beach Post, will join Kaiser Health News as a correspondent on June…

-
•
Appleby to report for Kaiser Health News
After 10 years at USA Today, where she covers the health care industry for the business section, Julie Appleby has…