Health Beat
Monitoring the Pulse of Health Care Journalism
Ad
-
How to care for your mental health over the next few years
A second Trump administration will likely make health reporters’ jobs more chaotic (again). Here’s how journalists — and a psychologist…

-
How a freelancer pitched a story on a controversial condition to Undark
For over a decade, journalist Jyoti Madhusoodanan wanted to write about Morgellon’s disease. She describes how she finally got the…

-
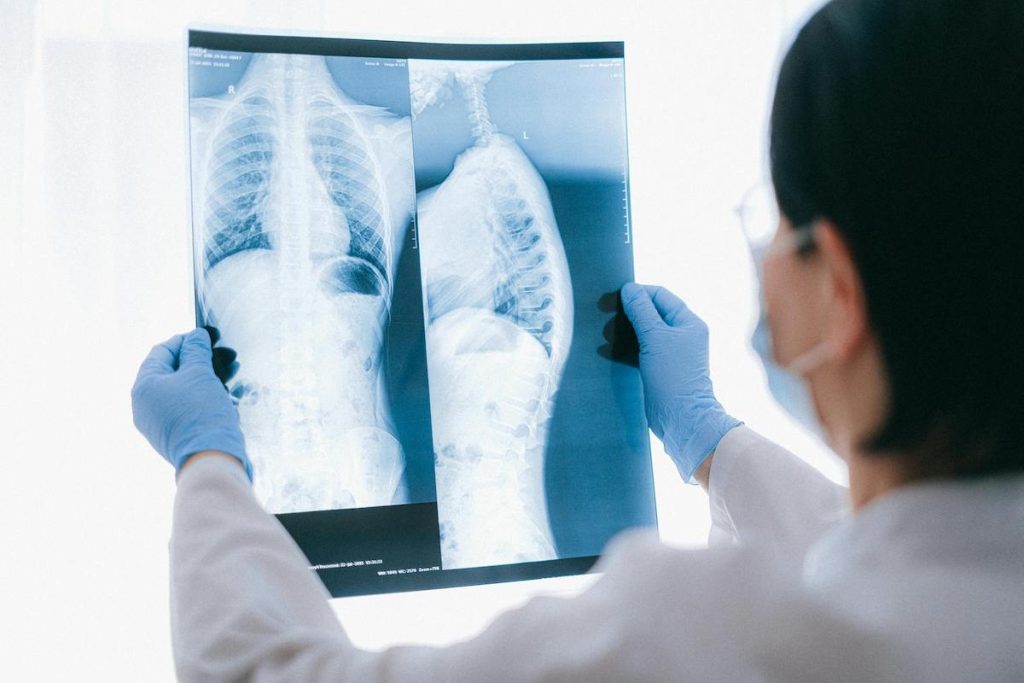
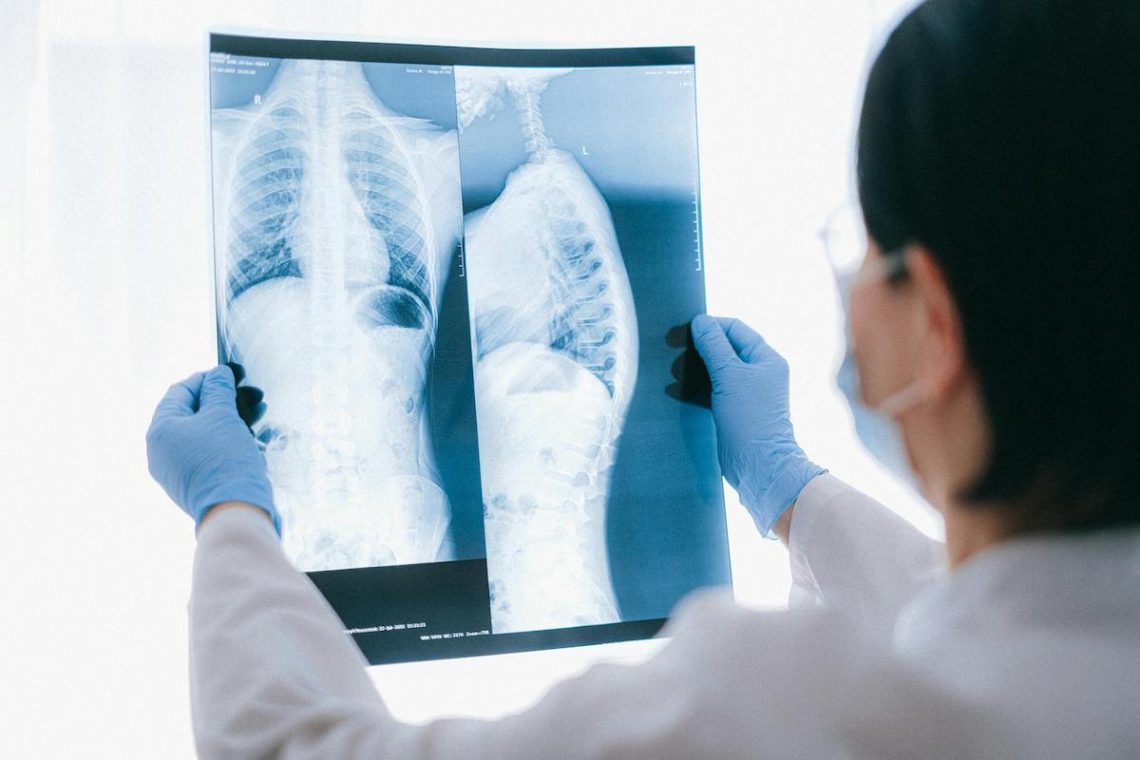
AI-generated X-rays stump radiologists: What does it mean for patient safety?
In a new study, only 41% of radiologists recognized that some of the X-rays they were reviewing were AI-generated.

-
How to cover the link between housing access and gun violence
Studies show that evictions, vacant homes and lack of housing access are closely connected to elevated gun violence.

-
Get credentialed with AHCJ’s first certification course
Test your knowledge and earn a certification with AHCJ’s new course, “Price and profits: Understanding the U.S. Health Care ‘System.'”

-
Race and risk in menopause: The connection journalists miss
Menopause is often discussed only in terms of hot flashes and mood changes — but research shows that it is a…

-
How to pitch editors right now
March’s Lunch and Learn revealed which best pitching practices are passé, and which help freelancers connect with editors now.

-
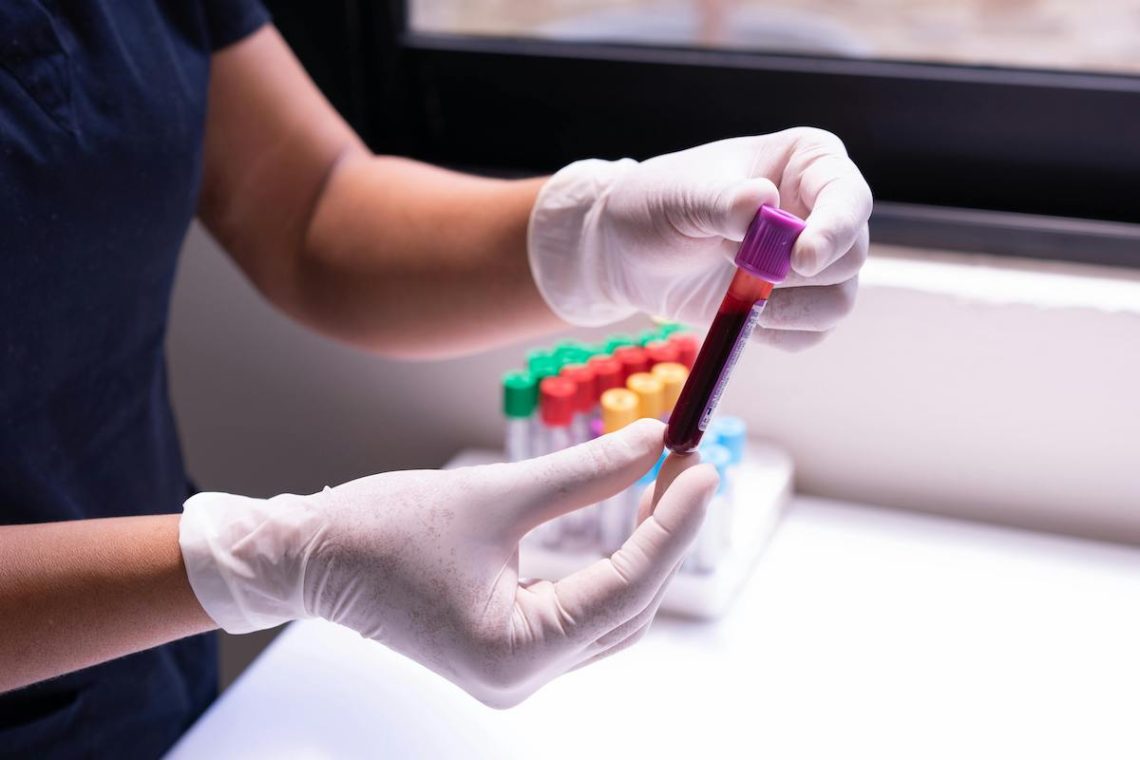
A simple blood test could detect Alzheimer’s earlier, but Medicare doesn’t cover it
Medicare doesn’t cover new FDA-approved blood tests that could detect Alzheimer’s early. The bipartisan “ASAP Act” aims to change that.

-
AI diagnostic risks top ECRI’s 2026 patient safety concerns
The list gives journalists a primer on current patient safety issues and can help identify emerging trends and generate story…

-
Scientists create ‘brain map’ targeting Alzheimer’s and other age-related diseases
A new study offers one of the most detailed looks yet at how the brain’s molecular landscape shifts over time…

Featured Resources
Work the Health Beat.
Get exclusive news and training opportunities from AHCJ in your inbox once a week.