Tag
lancet
-
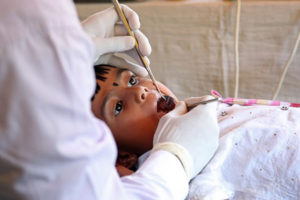
UN, medical journal highlight oral care as a global public health challenge
Painful and debilitating oral diseases such as tooth decay, gum disease and oral cancers are estimated to impact more than…

-
Researchers agree: A study is a terrible thing to waste
Spend any significant amount of time reporting on research and you’re bound to run across a real stinker of a…

-
The downside of using big data in medical research
A scientific scuffle played out in the pages of the Lancet recently. At issue was whether a team of scientists…

-
•
Critically evaluate, report on cell phone/cancer link
Courtesy of the International Agency for Research on Cancer’s press release and the storm of advance coverage it has spawned,…

-
•
International cooperative to share health data
Writing that “the importance of data sharing in advancing health is becoming increasingly widely recognised,” 17 major public health players…

-
•
BMJ: Wakefield’s vaccine-autism study fraudulent
The Internet and other media are abuzz with the news, published by BMJ yesterday, that the study published in The…

-
•
Journals pay for cracking down on industry funding
Paul Basken reports in the Chronicle of Higher Education that major medical journals, whose financial viability often depends heavily upon…

-
•
Journals pay for cracking down on industry funding
Paul Basken reports in the Chronicle of Higher Education that major medical journals, whose financial viability often depends heavily upon…

-
•
Journals pay for cracking down on industry funding
Paul Basken reports in the Chronicle of Higher Education that major medical journals, whose financial viability often depends heavily upon…

-
•
Journals pay for cracking down on industry funding
Paul Basken reports in the Chronicle of Higher Education that major medical journals, whose financial viability often depends heavily upon…