Several new blood-based tests can help physicians detect Alzheimer’s disease years before symptoms become debilitating, but because they’re considered screening tests, they’re not covered by Medicare.
This leaves millions of Americans undiagnosed or forced to wait until more serious symptoms appear. That means less time for early interventions, lifestyle and diet changes, or for anti-amyloid drugs to act more effectively.
Bipartisan legislation introduced in both chambers of Congress aims to change that.
The Alzheimer’s Screening and Prevention Act, or ASAP Act, would create a Medicare coverage pathway for FDA-approved blood biomarker screening tests, speeding up access to earlier diagnosis for the more than 7 million people in the U.S. living with Alzheimer’s. As many as half of those with the disease or at risk for it remain undiagnosed, according to University of Michigan researchers.
The legislation took center stage when its House sponsors, along with several clinicians, policy experts and patient advocates made their case at a March 26 policy forum hosted by The Hill and sponsored by the Alliance for Aging Research in Washington, D.C.. The bill was introduced by Rep. Vern Buchanan (R-FL), vice chairman of the House Ways and Means Committee, and Rep. Paul Tonko (D-NY); companion Senate legislation is sponsored by Senators Susan Collins (R-ME), Catherine Cortez Masto (D-NV), Shelley Moore Capito (R-WV), and Mark Warner (D-VA).
Why this Matters
The ASAP Act provides a good opportunity for journalists to speak with House and Senate co-sponsors about their positions on a variety of health funding, including: cuts to NIH-supported Alzheimer’s disease research; funding for programs supporting older adults; and how H.R. 1 might impact some Alzheimer’s related support services, like adult day centers, in-home care and caregiver respite.
Every year of delay has medical and economic consequences that compound the challenges of this disease, according to Rep. Tonko.
“I’m such a believer in science and evidence-based policy development. We need to commit to research,” he said at the forum. “We have a lot of investment to return to research which will help us along. Genomic discovery can assist in many areas of diseases that deal with the brain and the nervous system, because it’s less invasive, and because there is hope here to have earlier detection.”
The legislation addresses a basic gap in how Medicare covers preventive care. Under current law, Medicare can only cover preventive services specifically authorized by Congress or that are recommended by the U.S. Preventive Services Task Force. This process can delay coverage for years, according to Rep. Buchanan.
Medicare pays for biomarker blood tests only after a patient is already showing outward symptoms of cognitive decline. Screening a patient before symptoms appear is not covered.
“It is going to take an act of Congress,” said Joe Grogan, former director of the Domestic Policy Council in the first Trump administration and a non-resident senior scholar at the USC Schaefer Institute, speaking at the event. “CMS, if they just go through the normal process, will take years and years that we frankly don’t have. The ASAP Act is one of the most necessary pieces of legislation that I’ve seen.”
The blood tests
The FDA approved two blood-based biomarker tests in 2025 for people 55 and older who show signs or symptoms of cognitive impairment. One, designed for primary care settings, can rule out Alzheimer’s in those who test negative with a 90% negative predictive value. A second, used in specialty settings, can help confirm a diagnosis, reducing the need for spinal taps or PET scans that cost upward of $10,000.
Cara Leahy, D.O., director of cognitive disorders at Memorial Healthcare in Michigan, has ordered more than 400 blood biomarker tests over the past two and a half years. She describes them as transformative for her patients. “Within a week and a half, we know if we’re heading toward a diagnosis of Alzheimer’s or we’re looking in other directions,” she said. “It speeds that up incredibly.”
But nearly every time she orders a test, she also helps patients fill out a financial assistance form. Neither Medicare Advantage nor commercial insurance in her area covers the cost. For her largely low-income patient population, even $50 to $100 out of pocket can be a barrier.
Wider use of blood biomarker tests could help fill in some of the geographic gaps in care. In wide stretches of the country, including the Dakotas, Montana, Wyoming and Idaho, PET scans simply aren’t available, according to John Dwyer, president of the Global Alzheimer’s Platform Foundation. Even in major metropolitan areas, patients routinely wait 12 to 18 months to see a neurologist. “It doesn’t make sense,” he said.
The cost of doing nothing
The economic argument for early detection is mounting on multiple fronts. Georgetown University economist Carol Roan Gresenz, Ph.D., and colleagues found that financial red flags like declining credit scores, missed payments or maxed-out credit cards can surface as early as six and a half years before an Alzheimer’s diagnosis. Early detection could help patients and families protect themselves before those losses accumulate.
Globally, the cost of dementia care including foregone wages is projected to rise from $1.33 trillion in 2020 to $9.12 trillion in 2050, according to a 2024 study in npj Aging.
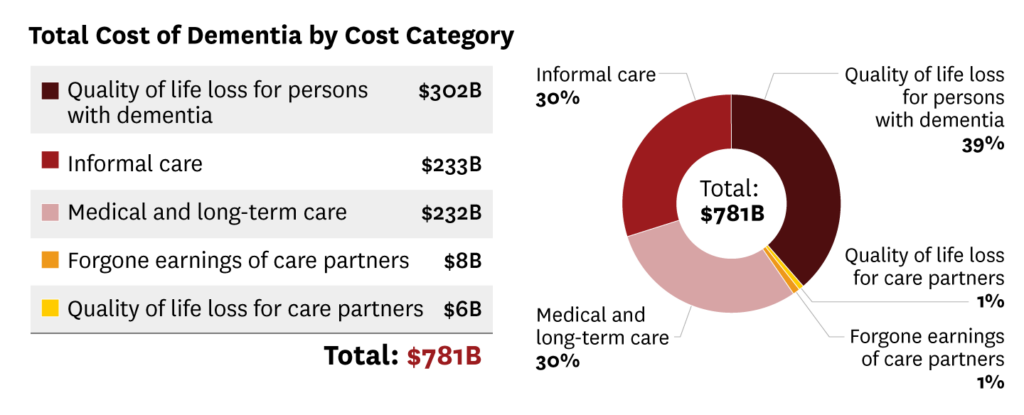
The U.S. spent an estimated $781 billion on dementia in 2025, according to the University of Southern California. This projection includes medical and long-term care costs, quality of life loss, caregiving hours, lost wages and more. Alzheimer’s already accounts for 20% of all Medicare spending; by 2050, analysts project it could consume one in three Medicare dollars.
“We can bankrupt the system, or respond with dignity and compassion,” Tonko said. “I would just question the people I serve with: Why is conquering Alzheimer’s not a high priority?”
The slow route from FDA approval to Medicare coverage has long frustrated patient advocates and health policy experts. Grogan, who also served in the George W. Bush administration overseeing HIV/AIDS policy, drew a historical parallel to those days.
“I don’t think this current debate is any different than we had at the dawn of the HIV epidemic, when there was a lot of pressure put on the government not to push screening for HIV because there weren’t effective treatments,” he said. “The same thing is happening now.”
A separate federal mechanism, Coverage with Evidence Development, was originally designed to speed new treatments into coverage while real-world evidence continued to build. However, it had the opposite effect, according to Grogan.
“CMS has used it to restrict access,” he said. “Some of these CED restrictions are in place for years and years. There are some that go on longer than a decade.” All of the currently approved anti-amyloid treatments for Alzheimer’s are subject to CED restrictions.
Funding cuts increase urgency
Even as the legislation gains traction, NIH funding for Alzheimer’s research has taken a sharp hit. The number of funded grants fell 52% between 2024 and 2025 — from 369 to 177 — according to Buchanan, who is retiring at the end of his term. He said he worries the cuts are eliminating the smaller, higher-risk grants that often produce the most original science. “That’s what we need now for Alzheimer’s,” he said.
The forum also featured a Q&A with Jay Reinstein, a former assistant city manager in Fayetteville, N.C., who received an early-onset Alzheimer’s diagnosis at age 57 after nearly 15 months of unexplained symptoms. He struggled to keep up during intense city council meetings: deferring to staff and working later hours, while chalking it up to information overload. He even bought a new BMW that he couldn’t afford, on a whim.
“If I was able to take this blood test earlier in my life, there’s no question that I would have made major adjustments,” Rhinestein said. “Diet and exercise are huge, and maybe thinking about a less stressful type of job,” he said.
The ASAP Act has approximately 50 co-sponsors in the House to date, and bipartisan support in the Senate. More than 459 leading organizations and researchers have urged Congress to pass the bill.
Resources
- New Blood Tests Can Detect Early Signs of Alzheimer’s: Should You Get One, (The New York Times, 3/19/2026)
- Determining eligibility for anti-amyloid treatments using blood biomarkers
- (Journal of Alzheimer’s & Dementia, December 2025)
- Alzheimer’s Association Clinical Practice Guideline on the use of blood-based biomarkers in the diagnostic workup of suspected Alzheimer’s disease within specialized care settings (Journal of Alzheimer’s & Dementia, July 2025)
- Alzheimer’s Disease Fact Sheet (National Institute on Aging)
- Understanding the new Alzheimer’s disease blood test and how it works (Mayo Clinic, November, 2025)